Magnetically Controlled Single Molecule Delivery
Funding Institution: Politecnico di Milano, grant 5×1000 junior – 2009
People: Prof. Matteo Cantoni (Project leader)
Partners: Prof. Ferrari Giorgio, Prof. Moscatelli Davide
Duration: 2 years (2011-2013)
The aim of this project is the realization of a system for the delivery of single nanobeads, loaded with biomolecules, to a desired location, e.g. a cancer cell, by a magnetic nano-dropper tool, based on the motion and trapping properties of magnetic domain walls.
Project Summary
The project investigates advanced systems for the manipulation, detection, and controlled transport of single magnetic nanoparticles in microfluidic and biological environments, with applications in biosensing and cellular interaction studies.
Bead detection using AMR sensors and impedance measurements
Two complementary detection approaches were explored: anisotropic magnetoresistance (AMR)-based sensing and impedance-based detection.
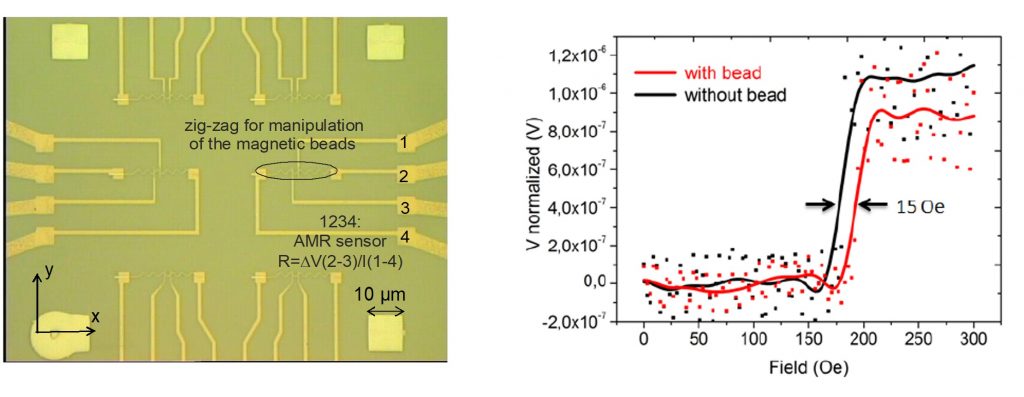
AMR sensors and domain wall manipulation
AMR sensors were integrated with permalloy zig-zag nanostructures fabricated using electron beam and optical lithography. The aim was to combine nanoparticle manipulation and detection within the same device by exploiting magnetic domain wall motion.
The devices consisted of permalloy nanostructures with Ti/Au electrical contacts and were protected with dielectric coatings (Al₂O₃, SiO₂, and Si₃N₄) to ensure stability in liquid environments. Experiments were conducted using 1 µm superparamagnetic beads in aqueous solution.
A stable anisotropic magnetoresistance signal of approximately 0.6% was measured. In addition, the presence of a bead induced a measurable shift in the domain wall depinning field of 15 ± 3 Oe, confirming successful coupling between the magnetic particles and the nanostructures.
The signal-to-noise ratio (SNR) was progressively improved from ~3 using standard commercial instrumentation to ~30 through the development of dedicated preamplification stages and custom lock-in detection electronics. These improvements enabled reliable detection of single-bead events in liquid conditions.
Impedance-based detection
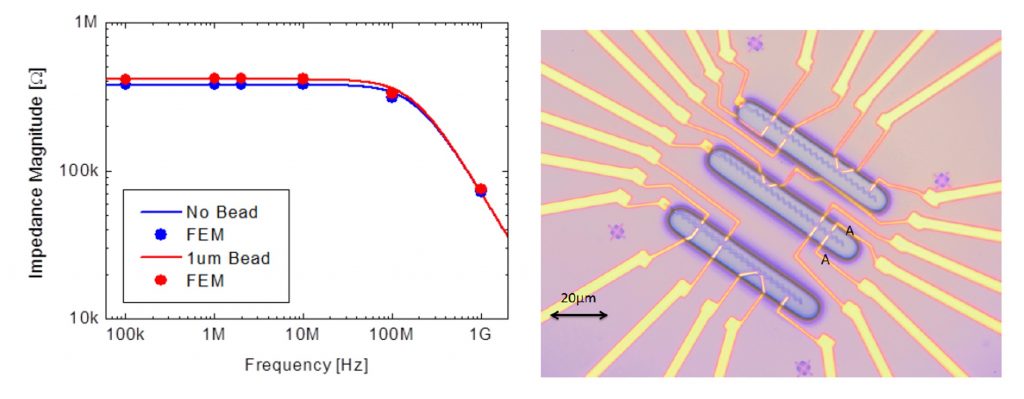
In parallel, an alternative sensing strategy based on impedance measurements was investigated. Planar electrodes integrated near the magnetic nanostructures were used to detect the passage of beads in a liquid medium.
Finite element method (FEM) simulations (COMSOL) confirmed that magnetic beads, being significantly less conductive than the surrounding phosphate-buffered saline (PBS), should produce a detectable impedance change when passing between electrodes.
Experimental validation was first performed using a macroscopic microfluidic setup, demonstrating measurable resistance variations consistent with theoretical predictions for beads of 1–2 µm diameter. These results confirmed the insulating nature of the beads and the feasibility of impedance-based detection at larger scales.
However, when implemented at the nanoscale, impedance sensors did not produce a clear or reproducible signal. Likely limiting factors included imperfect electrical insulation, suboptimal bead positioning relative to the sensing region, interfacial double-layer capacitance effects, and insufficient signal-to-noise ratio.
As a result, AMR-based detection was identified as the most robust and effective approach for single-particle sensing in the nano-dropper system.
Single-particle manipulation and cellular uptake
The final objective of this task was to achieve controlled delivery and manipulation of magnetic nanoparticles in proximity to living cells.
Magnetic ring structures and curved conduits were used instead of zig-zag geometries to enable smoother and more precise control of particle trajectories. Micromagnetic simulations were employed to optimize device geometry for maximizing magnetic forces acting on the beads.
This work was carried out in collaboration with biological partners, including IFOM and the Istituto di Ricerche Farmacologiche Mario Negri, which provided cell lines and experimental support, and Fondazione Filarete, which contributed surface coating materials.
Murine tumor cells were successfully cultured on patterned substrates (200 cells/mm²) and exposed to magnetic nanoparticles of 250–300 nm diameter. Surface functionalization and passivation layers (SiO₂, Al₂O₃/SiO₂, Al₂O₃/ZrOx) were optimized to balance nanoparticle mobility and cell adhesion in physiological media.
Experiments demonstrated stable nanoparticle manipulation for more than 5 hours under optimal conditions. Cellular uptake studies using confocal and fluorescence microscopy confirmed internalization of magnetic nanoparticles by epithelial human cells within approximately 4 hours under passive conditions.
Active manipulation experiments further demonstrated controlled delivery of nanoparticles in close proximity to both cancer and epithelial cells under physiological conditions, confirming the feasibility of magnetic guidance in biological environments.

Publications
M. Carminati, G. Ferrari, S. U. Kwon, M. Sampietro, M. Monticelli, A. Torti, D. Petti, E. Albisetti, M. Cantoni, R. Bertacco, “Towards the Impedimetric Tracking of Single Magnetically Trailed Microparticles”, Proc. SSD’14, 1569846595 (2014)
M. Monticelli, D. Petti, E. Albisetti, M. Cantoni, E. Guerriero, R. Sordan, M. Carminati, G. Ferrari, M. Sampietro, R. Bertacco, “Closed loop microfluidic platform based on domain wall magnetic conduits: a novel tool for biology and medicine”, MRS Proceedings 1686, Micro- and Nanofluidic Systems for Material Synthesis, Device Assembly and Bioanalysis (2014)
M. Monticelli, A. Torti, M. Cantoni , D. Petti, E. Albisetti, A. Manzin, E. Guerriero, R. Sordan, G. Gervasoni, M. Carminati, G. Ferrari, M. Sampietro, and R. Bertacco, “On-chip magnetic platform for single-particle manipulation with integrated electrical feedback”, Small 12, 921 (2016)
